
Significance of Blood Sample Collection and Recent Advancements
Blood draws play a crucial role in evaluating a patient’s condition and optimizing care during hospitalization. Approximately two billion blood tests are conducted annually in the United States [1], and laboratory test results inform 60 to 70% of medical decision-making [2]. While venipuncture is the most prevalent method for blood draws [3], vascular access devices (VADs), such as central lines, are also used for collecting blood samples [4] and are especially useful for patients with difficult venous access [4].
Blood draws are frequently performed for patient care and can lead to potential complications with important economic and clinical implications. During a hospital stay, patients experience around 1.6 to 2.2 blood draws per day, primarily through venipunctures [5]. Repeated needle sticks can result in multiple adverse events, including infections, nerve damage, inadvertent arterial puncture, vessel depletion, and hematomas. Beyond these clinical outcomes, repeat venipuncture may lead to significant pain and disrupt patients’ sleep and healing processes [5-7].
Complications may also arise in alternative approaches to blood collection, such as sampling from a central line. For example, use of central venous access devices can lead to increased hub manipulation and potential for intraluminal contamination, alterations in VAD patency, and erroneous laboratory values associated with the adsorption of medications infused through VAD [8]. More than 22% of daily central line access is for blood collection; therefore, minimizing these risks is critical to help improve care [4]. Industry studies have shown that decreasing the use of central lines for routine blood draws may reduce associated bloodstream infections [9], which represents an annual cost of over $2 billion to the US healthcare system [10].
The risk of pre-analytical errors is another important consideration with all blood draw methods. Hemolysis of blood samples can make the specimen unsuitable for laboratory testing [11], and may increase the probability of significant delays in care. Furthermore, these delays may lead to multiple redraws, lost labor hours, and delays in providing the right care to patients. Therefore, improvements in blood draw practices may help reduce the clinical/economic burden while enhancing patient/provider experience.
Recent advancements have enabled “needle-free” collection of blood specimens [12]. The PIVO™ Needle-Free Blood Collection Device (BD, Franklin Lakes, NJ), an FDA-approved medical device, connects to an indwelling peripheral intravenous catheter to help reduce the need for collection of blood samples with repeated needle sticks [3, 12]. The novel device provides high-integrity samples and equivalent laboratory results compared to traditional methods [13]. The device may also lead to clinical benefits and improved patient satisfaction, highlighting it as an attractive advancement for blood collection [12]. Due to its needle-less and user-friendly design, the device also has the potential to improve provider safety and prevent accidental needle sticks [13].
This case study provides an overview of the adoption of the PIVO™ device for blood draws at Virginia Mason Medical Center (VMMC). It examines the key processes and partnerships that led to the successful implementation of a novel device from the perspective of an early adopter.
Virginia Mason Medical Center
VMMC is one of the premier healthcare facilities in the Pacific Northwest. It is part of VM Franciscan Health, one of the largest healthcare systems in the Seattle-Tacoma area. VM Franciscan Health has 10 hospital sites serving acute care, behavioral health, and rehabilitation needs, as well as primary care and urgent care sites across the region.
The Previous State
At VMMC, there was a historic tendency to draw blood samples directly from a port-a-cath or central line, which originated from a desire not to inconvenience the patient with additional needle sticks. However, the repeated use of central lines was potentially contributing to high rates of central line-associated blood stream infections (CLABSIs). In fact, a particular unit at VMMC was driving CLABSI rates across the state, making reduction in CLABSI rates a priority for the medical center. One of the objectives of the new quality leader appointed in 2016 was to address this during her tenure. The impetus from the hospital leadership coincided with a national push to reduce hospital-acquired infections, considering their significant economic impact due to treatment with medications, increased length of stay, and other associated treatment costs [14].
The stakeholders at VMMC initiated their efforts with a root cause analysis to determine what factors may be contributing to CLABSI rates. The analysis highlighted a need for emphasis and training in appropriate and consistent blood draw practices. Therefore, the quality leadership developed a series of initiatives, including re-teaching the blood draw basics, reinforcing appropriate insertion/care and maintenance practices, as well as reviewing the reasons behind central line placement and ensuring timely removal. Additionally, the Infectious Disease team sought to reduce the frequency of blood draws from central lines, as other hospitals had done so to mitigate the risk of CLABSIs. While the appropriate use of a central line may not result in complications, certain factors related including those related to patient type, length of use, anatomical site of catheter insertion, and healthcare practice such as insertion practice and handling may increase risk of infection [15]; therefore, this step was included in the broader measures to reduce infections. Although CLABSI rates decreased following the implementation of these initiatives, the facility had ambitious plans to achieve further success regarding infection and contamination rates.
During their efforts to improve practice, the quality and nursing leadership identified the PIVO™ device at a conference and introduced the concept to other stakeholders at the hospital to receive their input. Initial impressions were positive, and the device was initially considered to replace venipuncture due to its potential for improving the patient experience and reducing repeated needle sticks. Eventually, the blood draws from central lines could also be transitioned to the PIVO™ device, representing a potential to reduce the incidence of associated infections and improve quality metrics. Based on these initial perceptions, the nursing leadership began the evaluation process for implementing peripheral line draws with the PIVO™ device.
Achieving Practice Change Through Stakeholder Engagement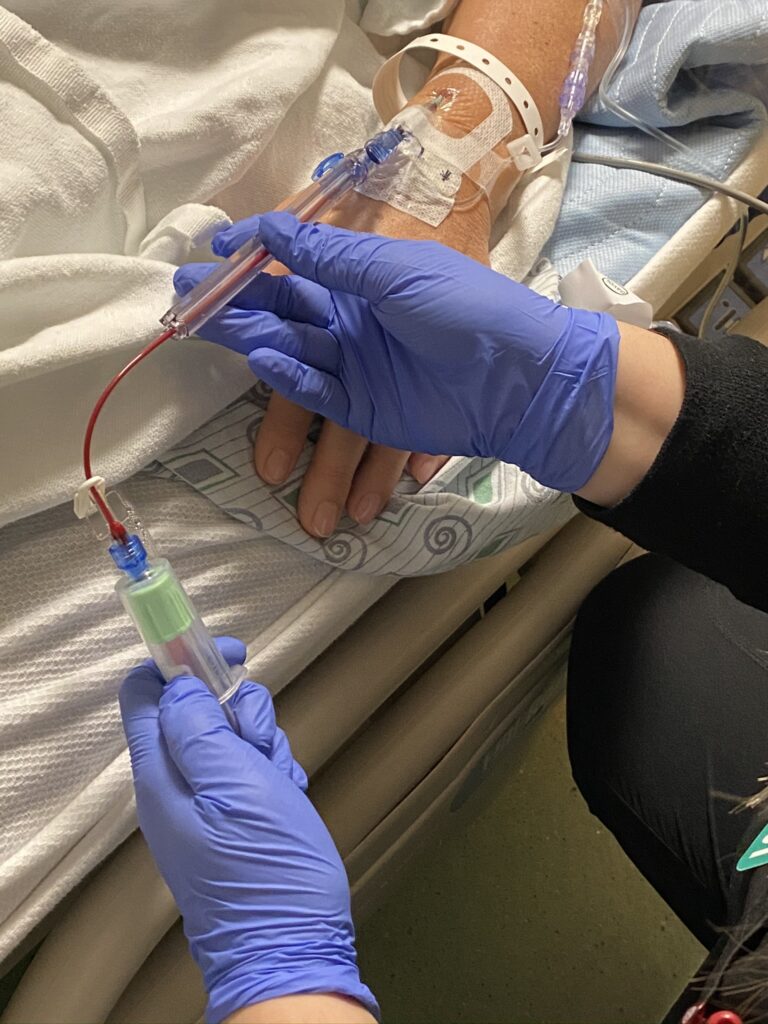
Following initial reception, stakeholders were eager to bring the PIVO™ device into VMMC and begin using the product first-hand. The implementation process began in 2018 and involved collaboration among multiple stakeholders across a variety of departments/functions, including the quality improvement and supply chain teams. Nursing leadership spent about 10 months preparing for the product evaluation to ensure a smooth adoption. Specific preparation efforts involved selecting the appropriate clinical sites, managing contracts/supply with the device manufacturer, and establishing protocols for implementation and associated data collection activities. Ultimately, three units were selected based on their high volume of blood draws: general med-surg, general surgery, and an additional surgical floor.
After these preparations, the physical implementation of the device started with the coordination between nursing and laboratory teams. As both teams would be the primary users of the PIVO™ device at the patient bedside and in testing the samples, their buy-in and onboarding were crucial steps for the success of the initiative. The implementation was also supported by various stakeholders that were involved in product evaluation, clinical integration, and outcome tracking processes. The Kaizen Promotion Office, which educates staff across all areas of the organization to facilitate change in culture and produce high-quality outcomes took part in understanding the relevant outcomes and data/metrics to assess the impact of peripheral line draws with the device post-implementation. This was crucial as the facility needed to collect its own data and confirm that the device was beneficial from both clinical and economic perspectives. The supply team contributed by stocking the device, making sure it could be implemented into current procedures, and planning the product evaluation. Lastly, the Scope of Practice Committee played a vital role to ensure smooth implementation by confirming that phlebotomists were allowed to perform peripheral line draws with the PIVO™ device. As their previous responsibility did not include drawing blood from a line, the scope of practice committee had to ensure that phlebotomists were authorized to perform peripheral line draws using the device prior to their onboarding.
Pre- and Post-Implementation Training
Given the novelty of the product, implementation required a comprehensive training program to ensure users were well-educated on the use and benefits of the PIVO™ device. Both the device manufacturer and hospital staff supported this training program, which included classroom training, bedside in-servicing and coaching, as well as ongoing training support.
From the beginning, the manufacturer offered on-site guidance and structured training to the internal VMMC education team. They provided educational modules for the VMMC personnel to complete before proceeding with classroom training, which consisted of a comprehensive explanation of blood draw procedures, why IVs have historically been unreliable for draws, and the benefits offered by the PIVO™ device. Following classroom training, the manufacturer performed bedside coaching and multiple two-week monitoring periods during which they observed live product use in the hospital. All new users were assigned educators who ensured proper blood draw technique was followed. The manufacturer then carried out post-implementation follow-ups and audits to focus their support on those facing challenges. They also offered a reference guide for clinical practice scenarios to assist VMMC educators in resolving any issues that arose during routine use. Furthermore, the manufacturer team remained available throughout the implementation process to provide any additional training as needed.
The VMMC staff provided ongoing education, through incorporation of PIVO™ device training into the new hire curriculum, and development of a long-term multi-phase program to ensure the sustainability of the correct practice. Although these intensive training programs were well suited to providers in high-volume settings, it was challenging for providers in lower-volume settings to reinforce required skills in their day-to-day practice. In one of these low-volume settings (i.e., the birth center), a nursing leader developed an additional training program to help new nurses, or residents, gain experience quickly. This program allowed new hires within the birth center to rotate, and complete shifts in higher-volume units or even travel with the IV therapy team to increase their exposure to peripheral line draws using the device. This approach was proven to be effective, as nurses returning to the birth center demonstrated improved skills and proficiency to utilize the PIVO™ device when needed.
Improving the Device Through Feedback from End-Users
As early adopters of a novel device, VMMC stakeholders, as well as other facilities trialing the PIVO™ device, provided feedback to the manufacturer to improve the stability and access of the IV when utilizing for draws.
Initial reports from the users were taken into consideration by the manufacturer, which led to the creation of a stabilizer component designed to help prevent the IV catheter pistoning/kinking, help reduce the compression of the underlying vessel, and help maintain the correct angle for attaching a flush syringe and connecting the PIVO™ device to avoid overmanipulating the catheter and creating vessel irritation or vessel spasms. The manufacturer incorporated all IV set-up components in a kit helping to streamline the process for users and supply personnel while optimizing the peripheral IV for compatibility, stability, and ease of use, improving the provider experience.
Implementation Hurdles and Solutions
Despite thorough planning and preparation, several challenges arose during the implementation process related to both clinical and economic evaluation. Challenges, such as those seen in this case, are common with the implementation of novel devices, and are inherent to driving a change in clinical practice. Each of these challenges provided an opportunity to better understand the implementation of new technologies such as PIVO™ in the hospital setting, and provide learnings for later adopters as well as new adopters of other novel technologies. Nevertheless, the VMMC team and manufacturer worked together to alleviate these obstacles and achieve long-term success. Many of these challenges were resolved as users became more proficient with continued use of the device and support from the hospital and the manufacturer, who worked to ensure that all stakeholders were comfortable with all aspects of the practice change.
Workflow Integration and Turnaround Times: As expected with a change in practice, the new line draw procedure initially resulted in an increased procedure duration. This was in part due to providers becoming acclimated to a new device; however, several nurses also highlighted there was an added step of using the PIVO™ device versus directly drawing blood from an existing central line. These concerns were largely resolved as practitioners became more comfortable with the device and observed potential improvements. Furthermore, the process became more efficient as providers were able to complete the blood draws quicker with more experience/training. As device utilization improved, clinicians, including both nurses and phlebotomists, also appreciated that they could perform blood draws conveniently at the bedside without the need for a central line, ultimately decreasing the total time to obtain a sample.
Training and Staffing Challenges: At the beginning of implementation, the understaffed state of relevant departments (i.e., core lab) resulted in delays in onboarding/training. This led to limited experience with the PIVO™ device and insufficient capacity to solve problems when needed. Training challenges were exacerbated by interruptions due to the COVID-19 pandemic, which introduced restrictions on the number of manufacturer representatives allowed in the hospital. Therefore, the burden of training fell to an already overextended hospital team during the pandemic. As restrictions were lifted, the manufacturer provided updated training sessions, along with a new fully integrated stabilized extension set, both of which played a major role in bolstering usage.
Achieving High Utilization: By the end of the product evaluation, peripheral line draws with the PIVO™ device were recommended as the preferred approach for drawing blood at VMMC, while using central lines or traditional venipuncture served as secondary options. However, particularly in clinically urgent situations, nurses or phlebotomists reverted to previous practice. As expected, there were also lower success rates on the first attempt blood draws during the training phase, leading to increased material costs due to additional attempts. As nurses and providers gained more experience, many began to prefer the new practice over traditional approaches as the benefits were recognized from both the provider and patient perspectives. Providers realized that patients were more comfortable with needle-free draws, and appreciated gaining new skills and responsibilities that the novel device offered. With experience, phlebotomists were grateful to learn a new method for drawing blood and to provide care with greater patient experience.
Demonstrating Cost Effectiveness: The primary economic obstacle was determining whether the device was cost-effective. This was a crucial hurdle since implementation incurred incremental costs for the hospital. In the initial phases, there were compounding material costs due to training, failed first attempts, and outdated/incompatible lines that could go unused. Department leaders and decision-makers wanted to know if the pricing was sustainable as the practice was expanding across the hospital. However, the utilization increased, and cost-related barriers were overcome by positive direct/downstream economic outcomes that were observed during the product evaluation period.
Observed Impacts of Peripheral Line Draw Practice Change
Several positive clinical, economic, and quality outcomes were observed by the providers and hospital leadership following the implementation of the PIVO™ device and other efforts to shift blood draw practices. Within the first year post-implementation, there were notable improvements in various metrics, including infection rates, hemolysis rates, and patient satisfaction.
Clinical Metrics and Workflow Efficiency: Overall, the stakeholders at VMMC reported reductions in hospital-onset bloodstream infections, TPA utilization for de-clotting lines, and overall central line days. As the majority of blood draw practice shifted to less invasive lines, central lines were not kept only for potential blood draws that may be needed. Furthermore, the practice change allowed nurses to draw blood conveniently at the bedside and share this responsibility with phlebotomists, which was especially important during the pandemic; fewer individuals were needed in patient rooms, resulting in decreased exposure/risk of COVID-19. Improvements in specimen quality and hemolysis rates were also noted as drivers of improved clinical efficiency and timely patient care.
Patient and Provider Satisfaction: Both providers and patients favored needle-free blood draws with the PIVO™ device. The patients requiring frequent tests were particularly happy to experience fewer needle sticks and a decrease in associated discomfort/pain. Moreover, the device and practice change allowed for better integration of care into the patient’s day since a sample could also be drawn in the morning by nurses who were performing other duties. Most providers were happy with causing less discomfort to patients, and some phlebotomists appreciated taking a more prominent role in patient care. Initial concerns with the learning curve associated with device use eventually disappeared, and the clinicians were pleased with the expansion in their scope of work, and their ability to improve patient experience.
Economic Considerations: While the full economic impacts of the implementation of the practice change were not captured, the savings from the practice change were clear through the reduction in adverse events and their associated costs. There were avoided costs associated with infection treatment, lab/materials costs of contaminated samples, lack of reimbursement or reimbursement penalties for hospital-acquired infections, and shorter hospitalizations. Worth noting, there were costs associated with implementation (i.e., obsolete extension sets), though these investments were necessary for improved blood draw practices.
Keys to Success in Achieving Sustained Utilization
The sustained use of the PIVO™ device, coupled with the observed clinical, economic, and patient satisfaction outcomes, indicate that the adoption of this novel device at VMMC was largely successful. Even in the face of challenges during the product evaluation process, VMMC and manufacturer stakeholders were able to deploy support and device iterations to promote the long-term success of the initiative. Understanding the crucial tactics and tools that led to success at VMMC can be illustrative to other organizations working to integrate a novel medical device into their workflow. Key contributors to success included the manufacturer’s dedicated involvement and obtaining stakeholder buy-in from the start.
Manufacturer’s Dedication to Improve Practice: The manufacturer’s commitment to enhancing patient care and patient/provider experience played a major role in facilitating successful practice change and ensuring the sustainability of its impacts. The clearest examples of this dedication can be seen in the significant contribution to training programs, and the iterations of the compatible IV set-up based on provider feedback.
The manufacturer was involved in training from the beginning, facilitating the initial orientation to the hospital education team to develop in-house expertise. The tools provided along with this orientation (e.g., reference guides) helped establish an ongoing collaboration in educational efforts. Beyond the physical tools, the 2-week evaluation periods conducted by the manufacturer showed not just their dedication to educating providers on the use of the product, but ensuring the appropriate technique and protocols as well as optimizing peripheral intravenous catheter (PIVC) practices. Ultimately, these educational efforts contributed to the comprehensive training programs developed by the internal teams at VMMC and achieving and sustaining positive outcomes.
A second example of the collaborative efforts between VMMC and the manufacturer was the open channel for feedback throughout the evaluation/implementation process. Through this collaborative approach, the VMMC stakeholders were given a unique opportunity to provide direct and thorough feedback and see concrete improvements in the experience of using the device and driving practice change. At the same time, the manufacturer received valuable insights into the real-world usage of the PIVO™ device, which led to the introduction of clinically informed components to improve patient care and provider satisfaction. The benefits of this process are not limited to VMMC and could impact patients and providers in various regions, as the adoption of this novel technology and related practice change increase across the nation.
Stakeholder Buy-In and Sustainability of Practice
Despite dips following initial implementation due to COVID-related challenges (e.g., personnel/stock shortages), line draws with the PIVO™ device were maintained at a consistent level. The current official hospital recommendations state peripheral line draws with the PIVO™ device as the primary method for blood draws. This was largely thanks to the efforts of VMMC leaders, who obtained buy-in from a variety of stakeholders from the beginning of the product evaluation and ensured that many perspectives were heard throughout the implementation process. Attention to the needs of the end-users, particularly with respect to training and device utilization, proved crucial to successful implementation.
Obtaining buy-in from key stakeholders was crucial to ensuring the successful adoption and sustainability of the practice. The goal for both VMMC leadership and the manufacturer was a long-term shift in blood draw standard of care. This is seen in part through the comprehensive training, which was integrated into the general hospital onboarding for new hires. Moreover, the ongoing training initiatives, including one-on-one preceptor sessions, hands-on training with dummy arms, and long-term support through the IV-therapy educator and manufacturer materials, illustrate a commitment to the proper technique and long-lasting positive outcomes.
Support for adopting the new technology and practice change is also achieved through the manufacturer’s openness to receive feedback and introduce additional components with an emphasis on the user experience. The introduction of the stabilizer and extension sets was largely focused on ensuring ease of use for providers. Rather than expecting providers to adjust to the product, the manufacturer developed and incorporated additional components to meet the needs of the clinicians.
Worth noting, the importance of stakeholder buy-in for lasting impact was highlighted by the variable levels of success across units. Units with more consistent and innovative training approaches generally observed greater success in implementation and associated improvements in outcomes, as evidenced by increased patient and provider satisfaction, higher device utilization, and greater reduction in infection/contamination rates. Moving forward, the observed positive impacts will likely lead to increased buy-in and interdepartmental learning to improve the blood draw experience for all patients. The VMMC leadership will continue to identify optimal clinical scenarios and determine the most appropriate utilization to enhance sustained use over the long term.
Final Thoughts
Novel and purpose-built devices can address longstanding and overlooked patient care challenges. VMMC’s experience with the PIVO™ Needle-Free Blood Collection Device demonstrates that novel solutions can be implemented successfully with a commitment to patient care and provider experience. The successful implementation of a new technology, and the sustainability of associated improvements, would not have been possible without the collaboration between VMMC stakeholders and the device manufacturer. With a solution-oriented approach, the VMMC team obtained buy-in from key stakeholders across the facility and implemented a new technology as part of a long-term quality improvement program based on the historic challenges identified by the hospital leadership. The device manufacturer kept an open channel to receive real-time feedback from the VMMC providers, improving the experience of using the device.
Ultimately, this collaboration enabled successful implementation for an early adopter of the device, despite challenges in changing a longstanding practice, ensuring proper training, and integrating a novel solution into the clinical workflow. VMMC stakeholders highly recommend that other institutions consider adopting new technologies, such as the PIVO™ device, and advise them to ensure that they have the right personnel, both internally and through partners, and obtain substantial buy-in from all parties involved early on. Lessons from VMMC’s experience as an early adopter of a new technology serve as guidance to other facilities and show that innovative solutions can address pervasive/overlooked issues and evolve the standard of care.
Authors:
Joy Selchow, MSN, RN, CRNI, Virginia Mason Medical Center, Seattle, WA, USA
Kimberly Alsbrooks, BSN, RN, RT (R), VA-BC, Becton, Dickinson, and Company, Franklin Lakes, NJ, USA
Shanna Salmon, MS-HCA, Becton, Dickinson, and Company, Franklin Lakes, NJ, USA
References:
- Balter, M.L., et al., Automated end-to-end blood testing at the point-of-care: Integration of robotic phlebotomy with downstream sample processing. Technology (Singap World Sci), 2018. 6(2): p. 59-66.
- Green, S.F., The cost of poor blood specimen quality and errors in preanalytical processes. Clinical Biochemistry, 2013. 46(13): p. 1175-1179.
- Pendleton, B. and R. LaFaye, Multicenter Study of Needle-Free Blood Collection System for Reducing Specimen Error and Intravenous Catheter Replacement. J Healthc Qual, 2022. 44(2): p. e24-e30.
- O’Malley, C.H., The frequency and reasons for central line accesses in critical care units. Canadian Journal of Infection Control 2018. 33(3): p. 165-167.
- Psaila, J., et al., Prospective Study Evaluating Whether Standard Peripheral Intravenous Catheters Can Be Used for Blood Collection Throughout Hospital Stay. J Infus Nurs, 2023. 46(1): p. 43-47.
- Matthews, E.E., Sleep disturbances and fatigue in critically ill patients. AACN Adv Crit Care, 2011. 22(3): p. 204-24.
- DuBose, J.R. and K. Hadi, Improving inpatient environments to support patient sleep. Int J Qual Health Care, 2016. 28(5): p. 540-553.
- Gorski LA, Hadaway L, Hagle ME, Broadhurst D, Clare S, Kleidon T, Meyer BM, Nickel B, Rowley S, Sharpe E, Alexander M. Infusion Therapy Standards of Practice, 8th Edition. J Infus Nurs. 2021 Jan-Feb 01;44(1S Suppl 1):S1-S224.
- Kuriakose, L., Decreasing Central Line Associated Bloodstream Infection Through Limiting the Use of Central Venous Catheters for Routine Blood Draws. J Dr Nurs Pract, 2020. 13(2): p. 173-183.
- Son, C.H., et al., Central line-associated bloodstream infection surveillance outside the intensive care unit: a multicenter survey. Infect Control Hosp Epidemiol, 2012. 33(9): p. 869-74.
- Lippi, G., et al., Hemolyzed specimens: a major challenge for emergency departments and clinical laboratories. Crit Rev Clin Lab Sci, 2011. 48(3): p. 143-53.
- Mulloy, D.F., et al., Effect of peripheral IV based blood collection on catheter dwell time, blood collection, and patient response. Appl Nurs Res, 2018. 40: p. 76-79.
- Cadacio, C. and I. Nachamkin, A Novel Needle-Free Blood Draw Device for Sample Collection From Short Peripheral Catheters. J Infus Nurs, 2017. 40(3): p. 156-162.
- Haddadin, Y., P. Annamaraju, and H. Regunath, Central Line Associated Blood Stream Infections, in StatPearls. 2023, StatPearls Publishing Copyright © 2023, StatPearls Publishing LLC.: Treasure Island (FL).
- Pitiriga V, Kanellopoulos P, Bakalis I, Kampos E, Sagris I, Saroglou G, Tsakris A. Central venous catheter-related bloodstream infection and colonization: the impact of insertion site and distribution of multidrug-resistant pathogens. Antimicrob Resist Infect Control. 2020 Dec 1;9(1):189. doi: 10.1186/s13756-020-00851-1. PMID: 33261661; PMCID: PMC7708904.
Articles you may like:
Clinical Integration for Your Value Analysis Program and Supply Chain

